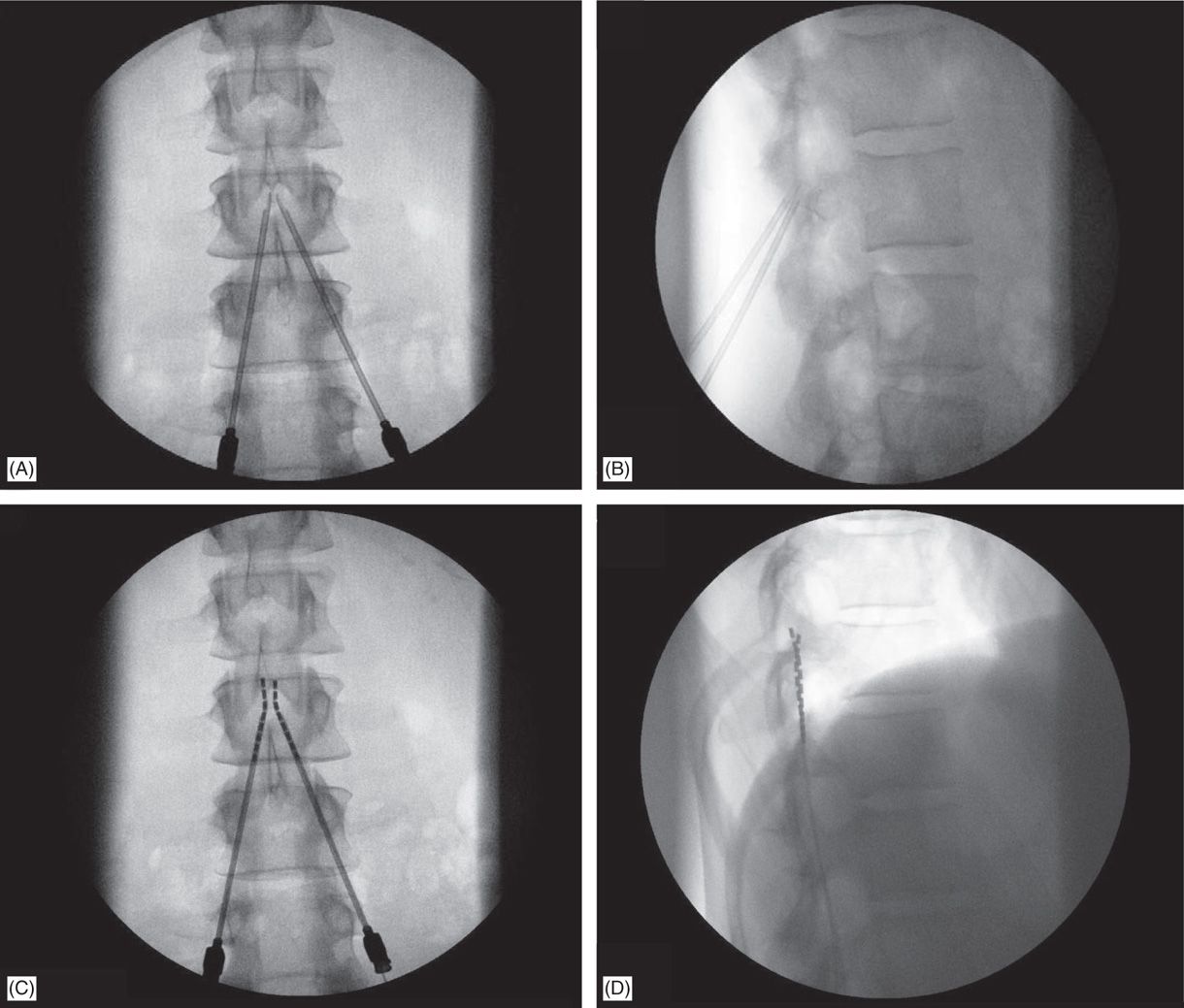 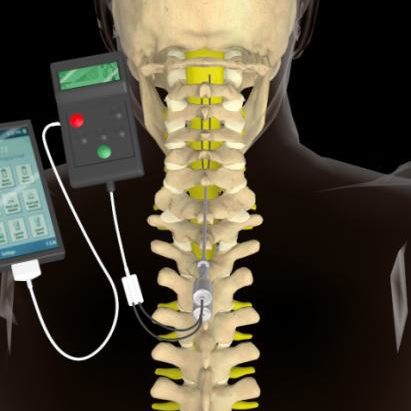 
Efforts to capture multidimensional aspects of chronic pain and treatment effects have historically been done through the addition of other validated PRO questionnaires 13, 14. The current gold standard for evaluating pain is using unidimensional PROs such as the Numerical Rating Scale (NRS) or the Visual Analog Scale (VAS) 11, 12. The condition of a person with chronic pain is usually evaluated through several patient-reported outcome (PRO) measures administrated manually in an in-clinic visit. Thus, appropriate individual selection and long-term monitoring are crucial in optimizing the outcomes of SCS therapy 9, 10. Response to therapy over time varies from person to person and often requires interactive adjustment of therapy parameters 8. Spinal cord stimulation (SCS) is an effective treatment option for chronic pain and often leads to pain reduction and improvement in quality of life 4, 5, 6, 7. Pain that lasts for more than three to six months is often considered chronic and is influenced by a complex combination of biopsychosocial factors including but not limited to emotional, psychological, physical, and social considerations 2, 3. Similar content being viewed by othersĬhronic pain is a debilitating condition affecting a widespread population in the United States, estimated at over 50 million American adults 1. The results of the study suggest that wearable biomarkers can be used to predict therapy outcomes in people with chronic pain, enabling continuous, real-time monitoring of patients during the use of implanted therapies. Feature importance analysis showed that digital biomarkers from the smartwatch such as heart rate, heart rate variability, step count, and stand time can contribute to modeling different aspects of pain. The model reached an accuracy of 0.768 ± 0.012 in predicting the pain intensity of mild, moderate, and severe. During the six months of the study, activity and physiological metrics were collected and data from 15 participants was used to develop a machine learning pipeline to objectively predict pain levels and categories of PRO measures. Twenty participants with chronic pain were recruited and implanted with SCS. This study aims to assess the feasibility of using digital biomarkers collected from wearables during SCS treatment to predict pain and PRO outcomes. Patient Reported Outcomes (PROs) from standard survey questions do not provide the full picture of what has happened to a patient since their last visit, and digital PROs require patients to visit an app or otherwise regularly engage with software. However, perceived treatment response to SCS therapy may vary among people with chronic pain due to diverse needs and backgrounds. All rights reserved.Spinal Cord Stimulation (SCS) is a well-established therapy for treating chronic pain. Benzinga does not provide investment advice. This article Back Off, Chronic Pain! FDA Nods for Abbott's Spinal Cord Stimulation Devices originally appeared on Price Action: ABT shares are down 0.12% at $109.71 on the last check Tuesday.ĭon't miss real-time alerts on your stocks - join Benzinga Pro for free! Try the tool that will help you invest smarter, faster, and better. On average, people who received SCS therapy experienced a 69.7% reduction in pain.īurstDR is a form of stimulation therapy that uses pulses-or bursts-of mild electrical energy without an abnormal sensation of "tingling" (known as paresthesia) to change pain signals as they travel from the spinal cord to the brain. ħ2.6% of people in the SCS study arm and 85.2% of those implanted achieved significant back pain reduction compared to only 7.1% in the conservative medical management arm.ĩ1.4% of people who received SCS therapy obtained significant pain relief or significantly improved function. 
Results at six months for the first 200 patients demonstrated clear clinical evidence supporting BurstDR stimulation therapy as an effective treatment for chronic back pain and were sustained at the 12-month follow-up.Īlso Read: Claims of PediaSure Boosting Children's Height Challenged in Lawsuit Against Abbott Laboratories. The FDA approved Abbott Laboratories (NYSE: ABT) spinal cord stimulation (SCS) devices for chronic back pain in people who have not had or are not eligible to receive back surgery, known as non-surgical back pain.Ībbott's DISTINCT study enrolled 270 participants who suffered pain for an average of 12.8 years.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
